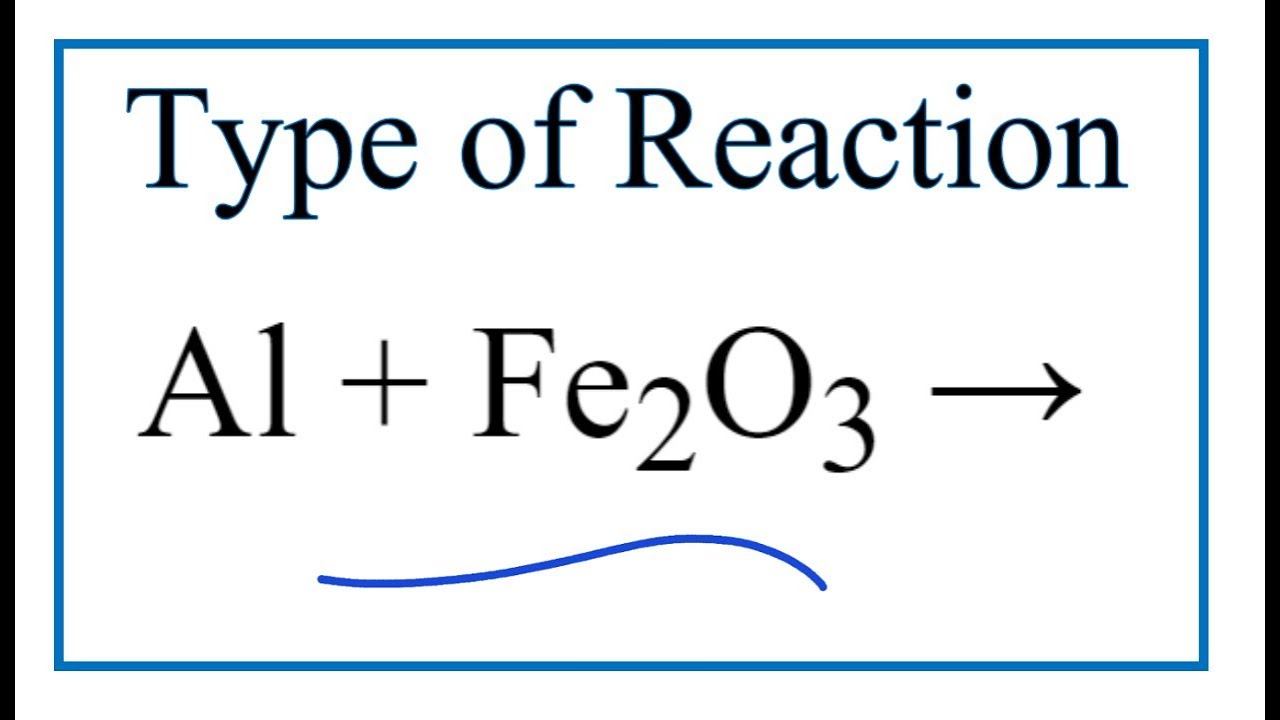Iron iii oxide react with aluminium and give molten iron and aluminium oxide write a balanced chemical - Brainly.in

Ferric oxide reacts with aluminium to produce aluminium oxide and iron The balanced chemical equation for the given reaction - Science - Chemical Reactions and Equations - 14356843 | Meritnation.com

Write the balanced chemical equation for the following reaction : "Iron II oxide reacts with Aluminium and gives molten Iron and Aluminium oxide."

SOLVED: Question 14 4 pts Given the following standard enthalpy changes of formation iron(Il) oxide; FeO(s) = -266 kJ/mol aluminium oxide; AlzO3(s) = -1676 kJ/mol aluminum; Al (s) = 0 kJ/mol iron,

Write the balanced chemical equation for the following reaction and identify the type of reaction and define it. 'Iron III oxide reacts with Aluminium and gives molten iron and aluminium oxide'.

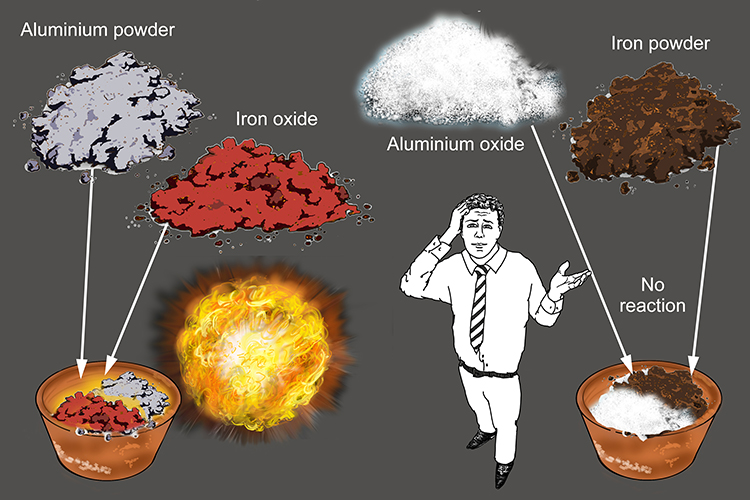
Thermit reaction, iron (III) oxide reacts with aluminium andgives molten iron and aluminium oxide. - YouTube

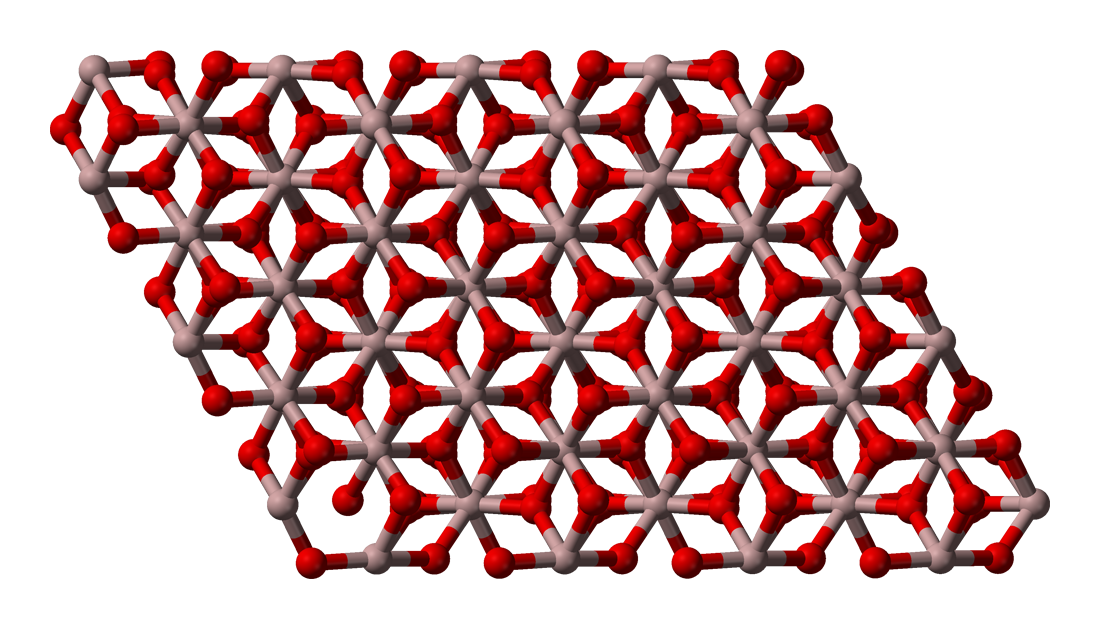
An extensive study on the synthesis of iron based magnetic aluminium oxide nanocomposites by solution combustion method - ScienceDirect
Is the reaction of Aluminium with any metal oxide(except iron oxide) during the thermite process exothermic or not ?